With the novel coronavirus continuing to disrupt millions of lives in the United States and countless more countries globally, it’s no surprise that vaccine manufacturers have reported unprecedented interest in volunteers looking to sign up for crucial phase 3 clinical trials. Both Pfizer (with partner BioNTech) and Moderna are looking to recruit 30,000 people each for testing their vaccine candidates, groups which will ultimately provide data on how effective the vaccines are in reducing the risk of coronavirus infection. (The Oxford vaccine, produced in partnership with AstraZeneca, is recruiting subjects in South Africa, the United Kingdom, and Brazil.) As a result of these trials, we may know as early as this year whether one or more vaccines could help prevent infections and the slow the pandemic.
Enrolling in a vaccine trial offers no guarantee of immunity to the novel coronavirus—half of those participating will receive a placebo shot—and it’s not simply one office visit. We’ve compiled some of the more pressing questions and answers to consider when deciding whether to join a trial.
1. Who is eligible to enroll in a coronavirus vaccine trial?
According to Meridian Clinical Research, which is helping to organize the phase 3 clinical trial for the Moderna vaccine, volunteers must be at least 18 years of age with no known history of coronavirus infection. The study’s organizers could also weigh factors such as being an essential worker, coming into regular contact with the public, or having underlying conditions like diabetes or high blood pressure. The trials are actively encouraging Black, Latinx, and Native American enrollees as well as older subjects in order to assess the efficacy of the vaccine in groups that are being hit the hardest by the virus.
The most important factor at this point may be the enrollment sites' locations. Meridian is currently recruiting subjects via nine offices in six states: Georgia, Louisiana, Maryland, Nebraska, New York, and South Dakota. Moderna uses several organizers and will operate 89 sites. Pfizer is looking for subjects in 39 states.
2. How do volunteers sign up?
Meridian Clinical Research is fielding volunteers for the Moderna trial via its website. Pfizer is inviting people to submit their requests online as well. To find a study in your area, you can also visit coronaviruspreventionnetwork.org, a site run by the National Institutes of Health (NIH). Potential volunteers enter some basic information in a brief questionnaire. If eligible, a representative will reach out to gather more information and set up enrollment.
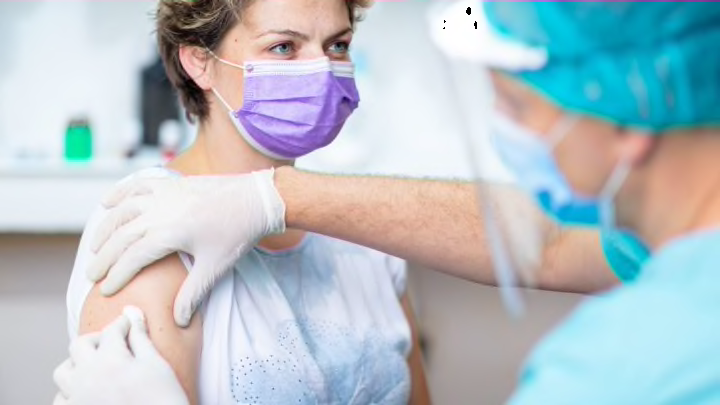
3. What happens when someone is accepted into a trial?
Those individuals deemed eligible to participate in a trial will be scheduled for an office visit where a baseline physical and bloodwork will be taken. They will also receive a COVID-19 test. Then, the participant will be injected with either a working vaccine or a placebo and monitored for side effects before being sent home. Both the Moderna and Pfizer vaccines are two doses, so subjects will need to return—typically in three to four weeks—for the second dose.
4. Are participants guaranteed to get an experimental coronavirus vaccine?
No. These phase 3 clinical trials are randomized, double-blind, placebo-controlled studies, meaning that half of the participants will get a placebo to create a control group. The results from this group will be compared to those who got the working vaccine. Neither the study subjects nor those administering the injection will know whether the injection had a working vaccine or the placebo, which is typically a harmless saltwater solution.
Even if the study subject receives the vaccine, there are no assurances it will provide any protection against coronavirus. That’s what the clinical trial is aiming to find out.
5. What are participants obligated to do?
Study subjects will be asked to commit to follow-up visits to assess their health and report any adverse effects. In the Pfizer study, volunteers will return a total of six times—including the initial two visits to administer the shot—over a two-year period. They may also be asked to keep a private online diary of symptoms.
6. Do volunteers get paid?
Generally, yes. As with many clinical trials, organizers will pay up to $2000 for subjects who complete the two-year observation period. The Pfizer study will also reimburse for expenses related to the study, like parking, meals, or other travel costs.
7. Are these trials safe?
By the time a vaccine begins a phase 3 trial, it’s already made it through the first two phases, which are focused on its safety in various doses. In fact, a vaccine is able to begin phase 3 and be administered to thousands of people because it demonstrated a promising safety profile in earlier stages. No major adverse events have been reported in the Moderna vaccine, which appears safe. The Pfizer vaccine has also demonstrated safety, though fatigue, headache, chills, or pain at the injection site have been observed for both vaccines.
It’s important to know, however, that a serious reaction is not out of the question. At least one participant in an earlier Moderna trial developed a fever of 103 degrees 12 hours after receiving a second dose and sought medical attention.
8. Is it possible to contract coronavirus from the vaccine?
No. There is no live virus used for either vaccine. Both the Moderna and Pfizer vaccines use a synthetic version of the virus's messenger RNA, or mRNA, to prompt human cells to produce protein from the virus that allows the immune system to mount a defense. The vaccination cannot infect anyone.
9. What happens if someone gets sick during a trial?
At no point is anyone involved in a phase 3 trial purposely infected with COVID-19. (That approach, called a “challenge trial,” is being heavily debated among scientists.) If a vaccine study subject becomes ill, they would likely be covered under their insurance plan. If the subject has no insurance and is harmed in some way as a result of the vaccine, then they may be eligible for financial reimbursement for medical costs. Because these policies can vary, it’s important for volunteers to request this information from the trial organizers and their own insurance company ahead of time.
10. When will researchers have an answer on whether the vaccines work?
Estimates vary widely on when data will be available to demonstrate how well the vaccines work in protecting subjects against COVID-19 infection. The studies need to enroll participants who meet eligibility guidelines, administer an initial dose, wait several weeks to deliver a second dose, and then wait to see who in the vaccine and placebo groups becomes infected. In order to assess the vaccine being 60 percent effective, for example, roughly 150 infections would need to be documented out of 30,000 people. Moderna believes an answer could come by November or December. Pfizer believes information might be available by October. The Food and Drug Administration has already stated a vaccine would need to be at least 50 percent effective to be approved.