Researchers funded by the National Institute of Biomedical Imaging and Bioengineering at Tufts University, and their collaborators, are the first to develop a 3D model to mimic human bone marrow that has successfully created blood platelets outside a human body.
Platelets, which are generated from giant blood marrow cells called megakaryoctyes, are necessary for blood to clot, and have been found to have a role in immune responses. They are generated in bone marrow, which fills the centers of bones. When bone marrow fails to produce enough platelets, the body will bruise and bleed more easily, blood won’t clot at cuts, and the chance of internal bleeding can increase.
To study microscopic, sensitive systems in the body like blood cells or bone marrow usually requires dissection or surgery, so researchers often turn to animal models before testing on humans. Bypassing animal or human subjects makes this 3D model especially exciting for researchers.
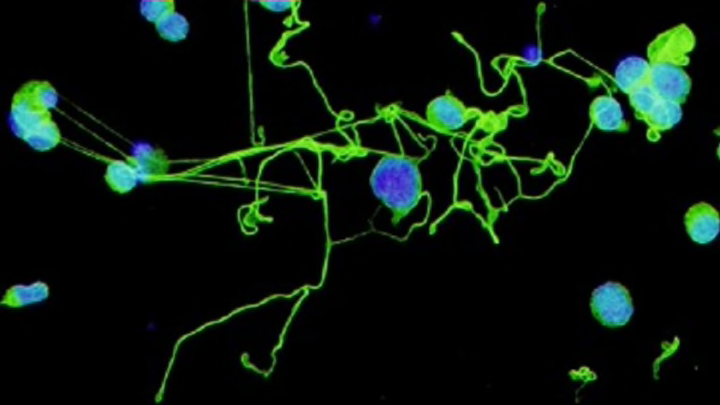
David Kaplan, chair of the department of biomedical engineering at Tufts, describes the environment they created to forge the platelets: “The megakaryocytes migrate through a spongy layer of silk that’s like bone marrow, hook up to the vascular silk tube they think is a blood vessel, send out long projectiles through the walls of these tubes, snip off the ends of those tubes and shed platelets into a flowing, artificial blood stream. Only if you put the right signals, morphology, and features together will the megakaryoctyes behave properly.”

Pbroks13, Wikimedia Commons
The cells were so convinced they were inside a human body, they shed new platelets. This was made possible by the versatile protein structure of silkworm silk.
The silks of silkworms and spiders are now being used regularly to create biomedical structures, particularly to mimic human tissue. They are incredibly compatible with human cells and easily customized to a wide variety of organic structures. “The silk itself is critical for our system because it gives us just the right chemistry and structure to avoid overstimulating the cells,” Kaplan says. “You can’t just use any material because you’ll prematurely cause platelets to aggregate."
The implications of this achievement are enormous for people who suffer from platelet diseases or who are undergoing chemotherapy. Kaplan tells mental_floss, “If we can develop a laboratory system to produce functional human platelets, you can imagine, as it gets scaled up and industrialized, you could have patients’ own systems generate platelets for them as needed. Right now you get them by transfusions, and matches are an issue.”
This 3D model method not only allows researchers to study human systems, it paves the way for research that can be done without animal testing.

David Kaplan, Tufts University
Medical biosilk has the potential to help create and study a wide range of human tissues for applications in numerous diseases. Kaplan also worked on a project using the biosilk to build a 3-D model of brain tissue. “You get real connectivity and can keep these tissues growing for extended periods of time, can look at structure, physiology and function and put it through a battery of tests,” he says. “For instance you can hit it with a hammer, mimicking traumatic brain injury and see how the tissue responds.”
He is also excited about the possibilities of new kinds of biomedical devices that can be implanted, such as silk mirrors. “This is a pure silk protein device that you can implant under the skin in a surgery, and when you shine light through the skin you get a higher intensity of reflected light back out. This is important if you want to use optics for diagnosis. It’s inexpensive, doesn’t hurt the patient, and degrades away without further surgery.”
From blood, to bone, to brain cells, when it comes to the possibilities for using biosilk in medical advances, Kaplan says, “We are just at the beginning.”